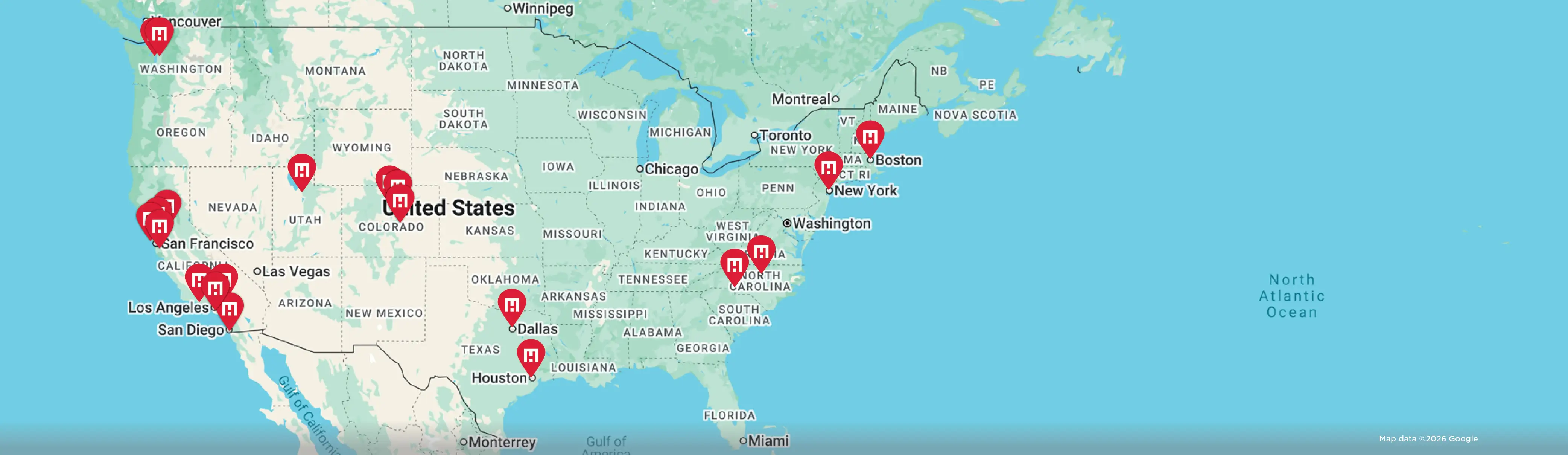
HM Science, the life science division of Hughes Marino, hosted two of the nation’s brightest minds in cancer research—Cigall Kadoch, Ph.D., Associate Professor of Medicine at the Dana-Farber Cancer Institute/Harvard Medical School and Investigator of the Howard Hughes Medical Institute, and Toni Choueiri, M.D., Director of the Lank Center for Genitourinary (GU) Oncology at Dana-Farber Cancer Institute and The Jerome and Nancy Kohlberg Professor of Medicine at Harvard Medical School—to discuss the most pertinent themes in cancer research today. Here is a recap of their discussion, which was moderated by Jarrett Collins, President of the Pan-Mass Challenge.
When you ask two of Boston’s leading oncology researchers about their professional challenges, it can be hard to anticipate how they might respond. After all, isn’t everything about their profession—striving to cure cancer—challenging?
In an intimate setting with other life science investors and executives at The ‘Quin House in Boston, Drs. Cigall Kadoch and Toni Choueiri brought the room to the edge of their seats. They humbly described how emotionally difficult it is to witness incredibly sick patients wait for new therapies. Kadoch reflected on her own impatience, “While we’re getting better at identifying targets and the mechanisms by which they promote cancer, the overall process of advancing medicines to patients is a very costly and time intensive endeavor.” Simply put, researchers feel the pressing need for maximum efficiency and it can be challenging to wait.
Dr. Choueiri echoed a similar struggle. While he thrives in the excitement of advancing research and witnessing his work save precious human lives, he is discouraged when he engages in hospice discussions. Despite extraordinary advances in cancer therapies, the discussion of end-of-life care still happens with youthful patients, even at the prestigious Dana-Farber Cancer Institute with worldwide leaders in oncology.
The room fell silent, quickly empathizing with the incredible burden Drs. Kadoch and Choueiri must carry as they navigate coming up with novel therapies. But perhaps this was the perfect segue to turn the audience’s attention to positive news—how far researchers have come in advancing cancer therapies. Drs. Kadoch and Choueiri shifted the direction of the conversation to the most promising research themes, and where biotech and pharmaceutical professionals, as well as investors, should hone their focus. Here are the six most impactful takeaways from their conversation.
This is a dream, but I believe that this dream is within reach. By the time my great grandchildren are adults, my hope is that cancer will not exist.
– Dr. Toni Choueiri

An understanding of cancer genomes has changed the landscape of oncology research.
According to Dr. Kadoch, we’re in a particularly exciting era, as merely a decade and a half or so ago, scientists began efforts to sequence cancer genomes. This helps researchers pinpoint the genetic alterations or mutations present in the very DNA of cancer cells and learn which mutations may be responsible for the development, progression, and therapy resistance features of human cancer. She firmly stated, “Genetics is the ultimate experiment and doesn’t lie. It tells us the hard-wired, contributing factors that underlie a given disease.” She continued, “Beyond informing the genetic makeup of a tumor, sequencing-based technologies can reveal, in a high throughput manner, the vulnerabilities of cancers, which can link genetic abnormality, tissue type, gene expression programs and other features to specific targets. Such targets may already have drugs developed against them, or, they prompt us to work toward developing them.”
Scientists are on the cusp of leveraging protein regions for therapeutic benefit.
According to Kadoch, about 40% of cellular proteins, including many involved in cancer, cannot be seen or visualized, meaning their functions are largely still mysterious even to leading bench scientists. This is particularly true for proteins in the nucleus of our cells, governing which of our 20,000 genes scattered across ~6 feet of total DNA are turned on and off. But there’s good news—the surges in cancer genetic sequencing coupled with new strategies for examining protein structures and resolving the functions of “unstructured” regions are beginning to surface and transition toward therapeutic evaluation in patients. Kadoch sees unique types of nuclear protein interactions in cells as promising areas of ongoing research with an eye toward drug development.
Researchers are pursuing synergy in cancer therapies, as well as refining biologic doses.
Dr. Choueiri emphasized that combining therapies is often the most effective approach for treating cancer. “This is how we cured TB. This is how we cured HIV, and this is how we cured some of the aggressive cancers, such as leukemia. It’s rare that one drug on its own works, depending even for tumors driven seemingly by one alteration in the DNA.” But now that therapies have been refined, and there have been great strides in how well they are tolerated by patients, physicians are able to combine them and achieve better outcomes.
In addition, Dr. Choueiri shed light on the advancement of biologic doses. Unlike traditional chemotherapy, which primarily aims to kill rapidly dividing cells, on-target biologic therapies work by interfering with specific molecular pathways or by harnessing the body’s immune system to fight cancer cells. Dr. Choueiri shared that researchers are enhancing dosing approaches for biologics, and these improvements can limit side effects for patients as well as increase the effectiveness of therapies.
Precision medicine has improved the type and dosing of medications for cancer patients, thereby limiting side effects.
Dr. Choueiri also discussed the incredible impact of precision medicine in treating patients. Advances in genomic and molecular profiling have enabled oncologists to tailor cancer treatments—be they different types or their timing and doses—to a patient’s specific tumor characteristics. Genetic testing and biomarker analysis help in identifying specific mutations or alterations that may predict response to targeted therapies. Overall, precision medicine is aiming to maximize treatment efficacy while minimizing side effects by selecting the most appropriate drugs and dosages for each and every patient.

Artificial Intelligence and computational biology have had a profound impact on cancer research—and will continue
to do so.
It is safe to say that researchers have more data at their fingertips than ever before. Empowered by the high-efficiency, parallel computing approaches central to AI, cancer datasets generated both locally and internationally can now be used for maximum benefit. As examples, such approaches can be used to detect cancer earlier—perhaps even at its earliest stages—and hence treat it more effectively. Just recently, machine learning models have been used to detect subtle abnormalities on routine imaging tests such as CTs and MRIs that might be missed by human radiologists, leading to earlier diagnosis and treatment.
In addition, AI can now play a crucial role in interpreting tumor genetic sequencing results from hundreds of thousands of tumors simultaneously and predicting their ramifications on cell behavior. This can help researchers use “gene and gene expression signatures” to predict treatment responses, and guide the selection of targeted therapies based on a patient’s individual tumor type—in essence, the concept of “personalized medicine.” It can also help researchers analyze the tumor microenvironment, including immune cell interactions, which is critical for developing immunotherapies and understanding tumor progression.
Finally, computational biology, including AI, can expedite the drug discovery process. AI models can predict how molecules will interact with specific proteins or inhibit cancer-related pathways, helping researchers identify potential drug candidates more efficiently.
The bottom line: There is incredible hope for the future of cancer research.
Drs. Choueiri and Kadoch left the audience with tremendous optimism for the future. They shared a vision for stamping out cancer through early identification and novel treatment strategies that grow directly out of academic research. Whether it’s by studying cancer genomes, dissecting specific regions of proteins, or by applying AI and computational biology to detect disease earlier and treat it more effectively—researchers have more powerful tools available now than ever before. For these Dana-Farber researchers, these tools and advances in research are made possible via financial support from the Pan-Mass Challenge, which has raised close to $1B for Dana-Farber Cancer Institute since its launch in 1980.
Support from the Pan-Mass Challenge is transformative. These unrestricted funds placed trustingly into the hands of our basic and clinical researchers inspire innovation and potentiate the discovery of new cancer therapies.
– Dr. Cigall Kadoch
In fact, research has taken such strides—and the future is so promising—that it could transform Dana-Farber Cancer Institute as an organization. Dr. Choueiri paraphrased his CEO Dr. Laurie Glimcherwhen he said, “In the next decade or more, Dana-Farber is not going to be where patients with cancer go. It’s going to be the place you go to so that you don’t have cancer.” Now that would be the dream, indeed.
 Cigall Kadoch, Ph.D., Associate Professor of Medicine at the Dana-Farber Cancer Institute/Harvard Medical School and Investigator of the Howard Hughes Medical Institute.
Cigall Kadoch, Ph.D., Associate Professor of Medicine at the Dana-Farber Cancer Institute/Harvard Medical School and Investigator of the Howard Hughes Medical Institute.

Toni Choueiri, M.D., Director of the Lank Center for Genitourinary (GU) Oncology at Dana-Farber Cancer Institute
and The Jerome and Nancy Kohlberg Professor
of Medicine at
Harvard Medical School.